Everyday we encounter salt, whether it be as a spice we use in our cooking, the salty winds off the ocean, the stinging of sweat in our eyes, the de-icer on the motorway or a multitude of other applications. But have you ever wandered what salt is?
If we take a deeper dive into the composition of salt, we find that in the practice of chemistry, salt is referred to as sodium chloride – a simple two element Ionic compound and is expressed using the formula Na+Cl-.
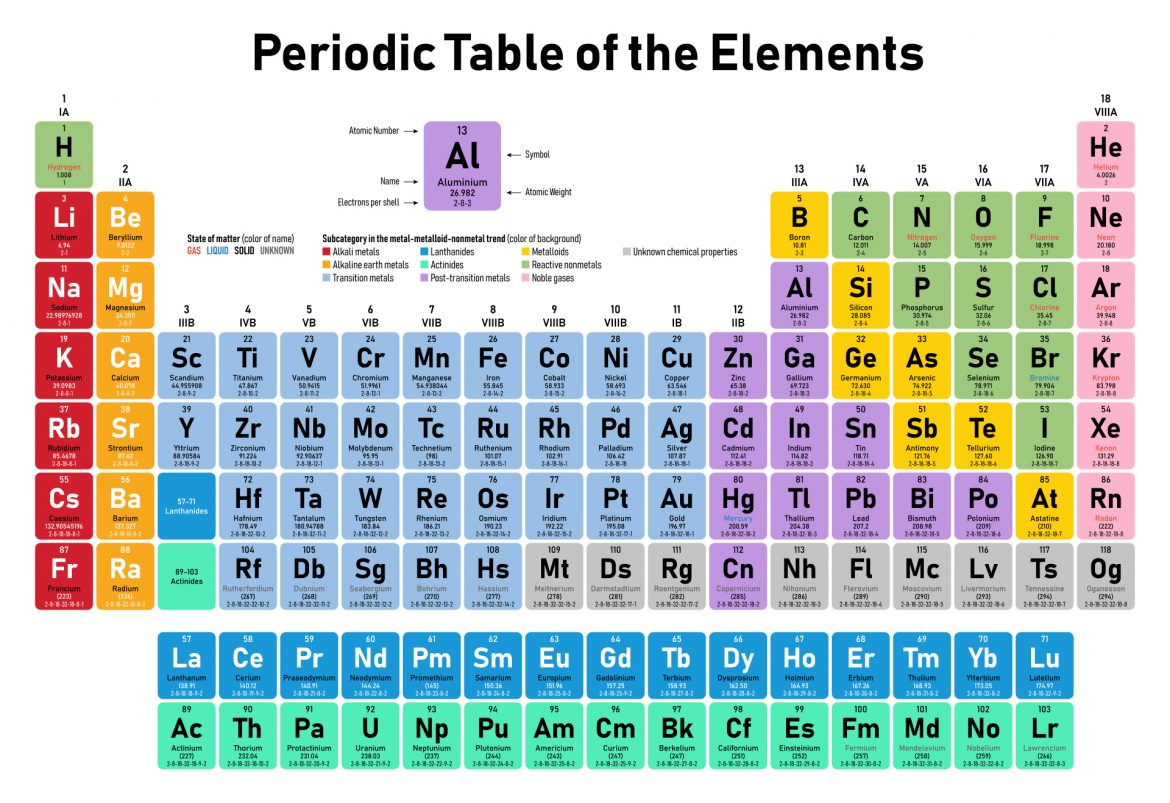
If we go back to our junior school science lessons, we will remember the infamous Periodic table of elements, which laid out for us the numerous symbols of the known metals, gases and other non-metals. This table reflects the properties of the elements in single state, in other words, they have not interacted with other atoms to form other chemical compounds.
So, when we refer to the formula of NaCl, we can start to understand how these elements came to become a unified crystal.
But let’s first refresh ourselves a little with some of the terminology used in chemistry.
- Compound -A chemical compound is any substance consisting of two or more different types of atoms or chemical elements
- Atom – an atom is the smallest constituent unit of ordinary matter. Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The atomic nucleus is made of one or more protons and a number of neutrons.
- Matter – any substance that has mass and takes up space by having volume
- Nucleus – the central region of an atom that consists of protons and neutrons
- Electrons – negatively charged particles that surround the atomic nucleus
- Protons – a fixed number of positively charge particles within the nucleus, that is equal to the atomic number
- Neutrons – a varying number of particles bound within the atomic nucleus and have a neutral charge.
- Chemical element – an atom that has the same number of protons in its atomic nucleus. This number of protons is equal to the atom’s atomic number in the periodic table.
- Atomic number – this is a unique identifier of an element that reflects the number of protons found in the nucleus of every atom of that element. In a neutral atom, this number will also correspond to number of electrons attached to the atom.
- Ionic Compound – a chemical compound that is held together by ionic bonds or Ions.
- Ion – an Ion is an atom whose bonds have been broken and reformed to leave a net electric charge (positive or negative), due to it having lost or gained one or more electrons. Once the neutral atom (ie equal protons to electrons) is no longer in equilibrium, it is said to have become an Ion. These Ions are then referred to as either positively charged (Cation) or negatively charged (Anion).
A lot to digest in all that, for which I apologise, but we can now pull these pieces together in getting a better understanding of what comprises Salt
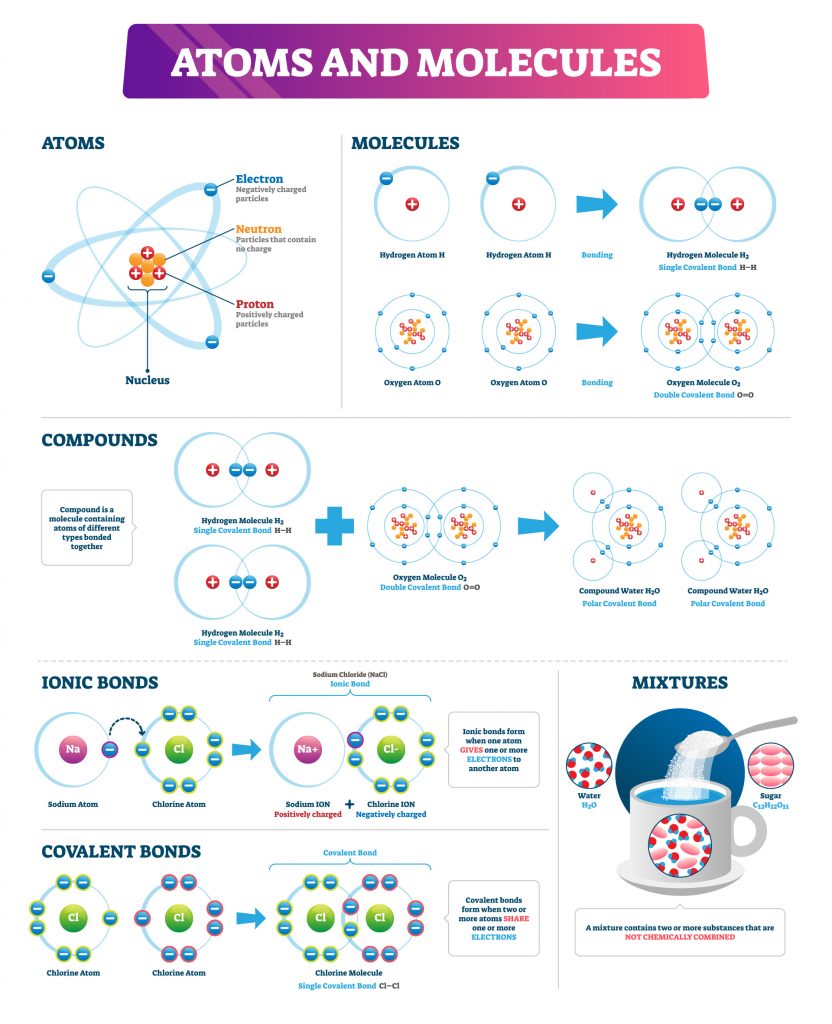
In the case of sodium chloride we see that the two elements, namely Na (Sodium) and Cl (Chlorine) have interacted with each other to form an ionic bond. The result of this interaction is that Na has lost an electron, therefore leaving it with a net positive charge, as there are now more protons than electrons. Conversely, Cl has gained an electron, leaving it with a net negative charge.
Hopefully that makes it a little more interesting now when you next sprinkle salt on your meal, you can visualize the various elements of that tasty little white crystal.